Sugars: from food industry to drug manufacturing
When it comes to sugar, people first think of its function as an energy supplier and sweetener in food or, in a negative context, of the common disease diabetes mellitus. But sugar is more than an energy supplier and nobody can live without it.
Most probably only a minority is thinking of a group of chemically similar molecules with diverse (bio)chemical functions.
Modification via glycosylation: dysfunction causes diseases
The glycosylation of certain proteins, meaning the addition of sugar moieties to proteins, is essential e.g. for correct protein folding and activity, whereas dysfunction of glycosylation is related to various diseases. Aberrant protein glycosylation for example is a hallmark of cancer and specific glycans actively drive tumor development and progression.
Thus, correct glycosylation is a critical attribute that can also be used to optimize known active ingredients. During development and administration, potentially active drugs routinely display intolerances or suboptimum therapeutic efficacies due to poor physicochemical and pharmacological properties in vivo.
Glycoengineering: slow release of active substances
Glycoengineering is reported as approach to improve active molecules, e.g. drugs or proteins, in terms of solubility, molecular stability, pharmacodynamic responses, and pharmacokinetic profiles (retardation effect). Besides, it is reported as strategy in cosmetics and perfume industry ensuring the slow release of the active substances over a certain period triggered by microbial or human glucosidases.
In addition, the glycosylation of known drugs may allow re-patenting of the new dosage form. One example in this regard is the modification of the well-known drug Paracetamol by the company 4GENE.
Key effects of glycosylation:
- Redesign of existing drugs
- Circumvention of existing patents
- Activity regulation
- Retardation effect and controlled drug release
- Solubility improvement
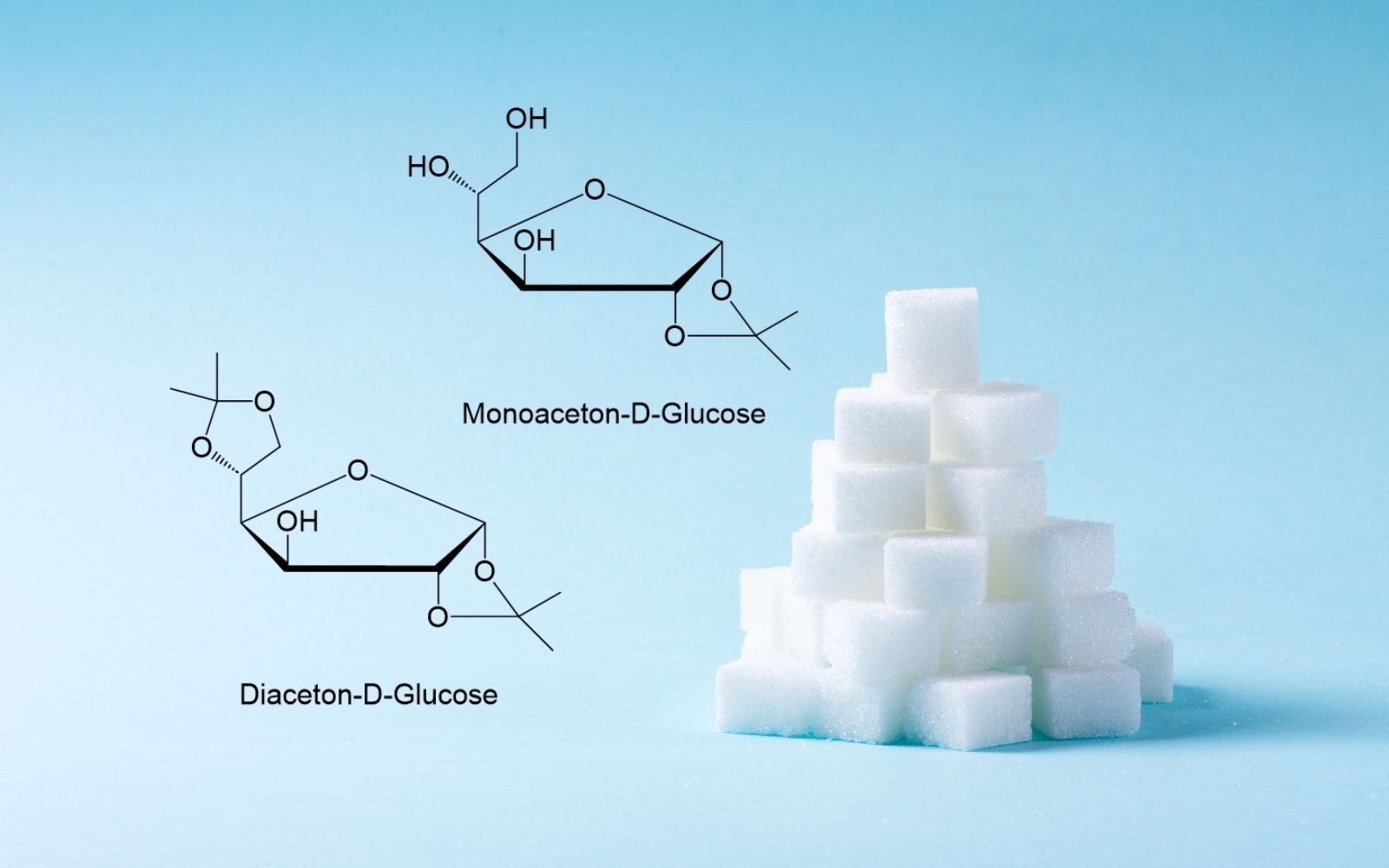
For carbohydrate chemistry and related chemical modifications, the commercial availability of the required (protected) sugar intermediates in sufficient quality is of crucial importance.
At Cfm Oskar Tropitzsch we have the capabilities to provide protected carbohydrates, e.g. Monoacetone D-Glucose (MAG) and Diacetone-D-Glucose (DAG) from 1 kg up to bulk quantities. The product is manufactured by our experienced joint venture partner. Consistent quality and delivery reliability are guaranteed.
Exemplary fields of applications:
- Chemical synthesis
- Pharmaceutical industry
- Metal chelators
- Glycosylation reactions
- Cosmetic industry
References:
PCT/US2009/035702
Synthesis, Biological Activity, and Molecular Modeling Studies of 1H-1,2,3-Triazole Derivatives of Carbohydrates as -Glucosidases Inhibitors; S. B. Ferreira, A. C. R. Sodero, M. F. C. Cardoso, E. S. Lima, C. R. Kaiser, F. P. Silva Jr., V. F. Ferreira; J. Med. Chem. 2010, 53: 2364-2375. https://doi.org/10.1021/jm901265h
Synthesis of polysulfated heparin degradation product; J. Kuszmann, G. Medgyes, S.Boros; Pharmazie 2004; 59: 344-348.
Development of a Flexible and Robust Synthesis of Tetrahydrofuro[3,4-b]furan Nucleoside Analogues; D. A. Candito, Y. Ye, R. V. Qziroz, M. H. Reutershan, D. Witter, S. B Gadamsetty, H. Li, J. Saurí, S. E. Schneider, Y.-h. Lam, R. L. Palte; J. Org. Chem. 2021; 86: 5142-5151. https://doi.org/10.1021/acs.joc.0c02969
WO 2010/036698 A1
US 2002/0173632 A1
Targeting Glycosylation: A New Road for Cancer Drug Discovery; A. F. Costa, D. Campos, C. A. Reis, C. Gomes; Trends in Cancer 2020; 6(9): 757-766. https://doi.org/10.1016/j.trecan.2020.04.002
A new alternative tool to analyse glycosylation in pharmaceutical proteins based on infrared spectroscopy combined with nonlinear support vector regression; S. Hamla, P.-Y. Sacré, A. Derenne, K.-M. Derfoufi, B. Cowper, C. I. Butré, A. Delobel, E. Goormaghtigh, P. Hubert, E. Ziemons; Analyst 2022; 147: 1086-1098. https://doi.org/10.1039/D1AN00697E
Glycosylation and Lipidation Strategies: Approaches for Improving Antimicrobial Peptide Efficacy; R. Bellavita, S. Braccia, S. Galdiero, A. Falanga; Pharmaceuticals 2023; 16(3): 439. https://doi.org/10.3390/ph16030439
Glycosylation of Therapeutic Proteins: An Effective Strategy to Optimize Efficacy; R. J. Solá, K. Griebenow; BioDrugs 2010; 24(1): 9-21. https://doi.org/10.2165/11530550-000000000-00000
4GENE https://4gene.de/




 4c media
4c media